- Home
- Herbal Remedies
- Caprylic Acid
Does Caprylic Acid Work for Yeast Infections?
Posted 6/15/2025
Written By Pharmaceutical Scientist Dr. Harmeet Kaur, PhD
Caprylic acid (CAP) (from the Latin term Capra, meaning "goat"), also named as octanoic acid is a medium-chain saturated fatty acid (MCFA) with the structural formula CH3(CH2)6CO2H. It is a colorless oily liquid with a slightly unpleasant rancid-like taste and smell, and have negligible solubility in water. It is found as a constituent of coconut oil and palm kernel oil and naturally in the milk of various mammals including man.
Caprylic acid does not need bile acids or lipases for absorption. After reaching the intestine, the acid moves directly to the liver via the portal artery, making it a readily available energy source for the human to efficiently utilize and less likely to be stored in form of fat.
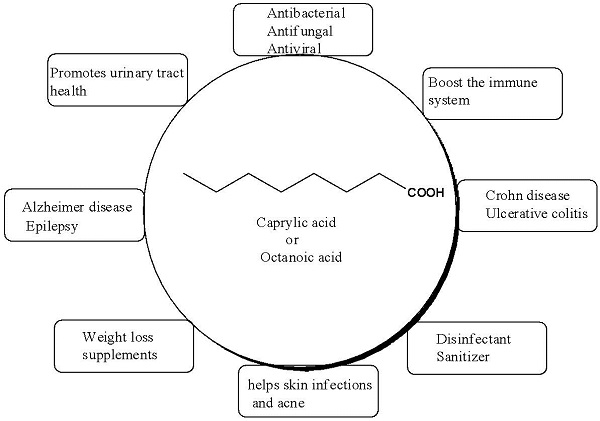
It is an antimicrobial lipid that is referred to as a single-chain lipid amphiphile that can interact with bacterial cell membranes and exhibit antimicrobial activity. It is also part of many dietary supplements, claimed to have anti-inflammatory and antimicrobial properties. It is the chief constituent of medium-chain triglyceride (MCT) oil, a dietary supplement assumed to assist in weight loss and athletic performance, and also a popular ingredient in several anti-aging creams [1,2, 3].
How Does Caprylic Acid Work?
It is an amphipathic molecule like the phospholipid bi-layer of the cell membrane, with the hydrocarbon chain constituting the hydrophobic part while the carboxylic acid group comprises the hydrophilic part. At pH above 4.8, this fatty acid occurs in the deprotonated or anionic form. Hence in most physiological environments of the body, the caprylic acid is rapidly ionized to a benign nutrient.
Mostly topical formulations presented protonated caprylic acid which retains antimicrobial activity by having a pH below 4.8. Also, the natural acidic pH range is seen in human skin, epithelial environments such as the digestive tract, and the vaginal canal.
The monoglyceride form of fatty acid which comprises of a fatty acid linked with a glycerol molecule by an ester bond differs by not having ionizable proton across relevant pH conditions, and hence, present nonionic molecules with neutral electrical charge and a certain degree of polarity.
Due to structural similarity with biological membranes, the fatty acids and their monoglycerides can insert themselves into the bi-layer of the membrane results in pores in the membrane followed by disruption of the normal processes of the membrane and hence impacts cell permeability. The pores permit for leakage of the intracellular contents, which compromises the cell’s integrity leading to lysis of the cell followed by eventual cell death [4].
Does Caprylic Acid Work as an Antibacterial Agent?
Several studies have been reported the effects of saturated fatty acids (with a chain length of 2 to 18 carbons) on Gram-negative bacteria, including Salmonella species and Escherichia coli, and demonstrated that caprylic acid has predominantly high antibacterial activity against these bacteria [5,6].
Skrivanova et al evaluated the in vitro susceptibility of Salmonella spp. (S. enteritidis, S. infantis and S. typhimurium), to 15 fatty acids in cultures grown on glucose. Caprylic acid was found to be the only acid that inhibited glucose utilization with IC50 ranges from 0.75 to 1.17 mg/mL against Salmonella spp [7].
Kollanoor and his colleagues also showed the antibacterial activity of the caprylic acid and its monoglyceride, monocaprylate, against fish pathogens including Edwardsiella species [8].
Caprylic acid and monocaprylate have also shown potent inhibitory activity towards main foodborne pathogens, as well as E. coli O157:H7, along with Salmonella species, Listeria. monocytogenes, and mastitis pathogens such as Streptococcus dysgalactiae, Streptococcus agalactiae, Streptococcus uberis, and Staphylococcus aureus [9].
Mayri et al reported that caprylic acid (0.8% v/v) in combination with rhamnolipids (0.2% v/v) or sodium dodecyl sulphate (0.2% v/v), inhibit the growth of Bacillus subtilis and Staphylococcus aureus. Also, caprylic acid (0.8% v/v) in the presence of sophorolipids at 1% v/v inhibited the growth of E. coli, P. aeruginosa, B. subtilis, and S. aureus [10].
Caprylic acid, monocaprylin, and sodium caprylate, also proven to be potential substitutes for traditional antimicrobials against Dermatophilus congolensis (Gram-positive bacterium) which causes skin disease in a wide range of animals including man. Caprylic acid exhibits its antibacterial action on D. congolensis by disrupting the plasma membrane and obstructing many cellular pathways by inhibiting the production of RNA polymerase B, superoxide dismutase, and heat shock proteins [11].
Caprylic acid is also found to decrease the extent of swelling and pustules in mice infected with Propionibacterium acnes, a bacterium that is involved in the pathogenesis of acne [12].
Cronobacter malonaticus and Cronobacter sakazakii are infections causing pathogens in children mainly linked to the ingestion of contaminated milk and food. The treatment with caprylic acid and monocaprylin at 2 mg/mL (temperature 40–55°C) reduce the no. of viable cells by five times in C. sakazakii DBM 3157T and below limits of detection in the case of C. malonaticus DBM 3148. The monocaprylin exerts its action by the release of cellular proteins and nucleic acids followed by damage to cytoplasmic structures resulted in cell aggregation [13].
Kim et al demonstrated the combined treatment with low concentrations of caprylic acid and citric acid, at mild heating temperature (45°C or 50°C) will produce a synergistic effect and can eliminate virulent pathogen E. coli O157:H7 and the endogenous microflora from unpasteurized carrot juice and hence can be used for the preservation of unpasteurized and fresh vegetable juices [14].
Does Caprylic Acid Work for Yeast & Other Fungus?
Several reports revealed the potential of saturated fatty acids (2 to 18 carbon atoms), for the inhibition of growth of certain molds, such as Aspergillus niger, Rhizopus nigricans, Aspergillus glaucus, Penicillium frequentans, etc.
Tohru Tasukahara demonstrated the greatest fungistatic activity for caprylic acid among all the normal saturated fatty acids (C2 to C18) studied against C. albicans at pH 5.91. It was fungistatic in a concentration of 1/1,600 M [15].
Neuhauser and Gustus reported that a therapeutic agent, consists of saturated complexes of an acid-adsorbing resin and caprylic acid, seems to be successful for the treatment of severe intestinal candidiasis. The caprylic acid-resin complex exerted an excellent inhibition of the growth of C. albicans in vitro [16].
Later on, the acid was found as a fungicide rather than as a fungistat against C. albicans. The fungicidal activity of caprylic acid depends upon several factors such as the concentration of the acid, the period of contact, and the pH of the media.
The caprylic acid fungicidal activity is considerably reduced by the simultaneous addition of several agents like a serum, polyhydric alcohol, and carbohydrate. The disappearance of this protective effect, after the contact with the acid for 3 hr suggested the strong and specific adsorption of caprylic acid to the yeast cells.
Caprylic acid exerts its antifungal action against Candida by interfering with morphogenesis (a process in which Candida can change from its standard yeast form to the more infectious hyphal form), affecting the adhesion properties of Candida, and biofilm formation [17].
The combination of natural antifungals like caprylic acid with other natural ingredients creates a more potent, multi-faceted approach to killing off Candida yeast, in addition to decreasing the chance for the yeast to adapt to a single agent.
Yoon Seol and Min Suk found that the combination of caprylic acid with thymol and carvacrol at low concentrations had synergistic fungicidal effects within short durations against C. albicans. The synergistic mechanism may involve the increased entry of the antifungals into the cells due to the disruption of the fungal membrane, primarily caused by caprylic acid, and by inhibition of the efflux of antifungals by caprylic acid, thymol, or carvacrol [18].
Caprylic Acid Helps Remove Biofilms
Biofilms are three-dimensional dense colonies of microbial cells attached to the surfaces that are enclosed in shielding biopolymer matrices secreted by the embedded microbes. These microbial biofilms have been reported to be associated with a significant proportion of human microbial infections.
Joel Rosenblatt et al demonstrated the rapid and complete biofilm eradication in an in vitro model with synergistic combinations of glyceryl trinitrate and caprylic acid against clinical isolates of methicillin-resistant Staphylococcus aureus (MRSA), methicillin-resistant Staphylococcus epidermidis (MRSE), multidrug-resistant Pseudomonas aeruginosa, and C. albicans, representative of key Gram-positive, Gram-negative, and infectious fungal pathogens [19].
In a further study, the authors reported the complete eradication of microbial biofilms with synergistic combinations of caprylic and polygalacturonic (PG) acids within 60 min in an in vitro biofilm eradication model against the representative foodborne and hospital infectious pathogen (MRSA, multidrug-resistant Pseudomonas aeruginosa, C. albicans, E. coli, and S. enteritidis).
Caprylic acid at 0.1 and 0.4 % concentrations has shown a decrease in viable MRSA and C. albicans organisms in biofilms after 60 min. incubation, however, in the case of P. aeruginosa biofilms complete eradication was seen within 60 min.
The combination of acids reveals its safety for normal cells by not inducing any cytotoxic response in mammalian fibroblasts. This along with the prior history of safe use of the individual components in nutrition applications and wound healing suggests that the combination should be suitable for topical and food disinfection uses.
A hypothesis for the higher synergy with caprylic acid in combination with PG may be that the apparent solubility of caprylic acid is enhanced in the combination and the bioavailability of both caprylic acid and PG within biofilms is expressively enhanced.
The dual mechanisms of antimicrobial synergy for caprylic acid and PG combinations have been hypothesized. The protonated caprylic acid has known to directly interrupt cell membranes while PG is capable of binding or precipitating significant metal ions and molecules with cationic residues like peptides and proteins resulted in inhibition of microbial proliferation and survival, predominantly when cell membranes are concurrently stressed and become more porous by the action of protonated caprylic acid [20].
Caprylic Acids Other Uses
Caprylic acid supports a healthy gut and aid in the control of inflammatory bowel diseases (IBD), including Crohn's disease and the characteristically more severe ulcerative colitis. It is believed to suppress the production of an inflammatory cytokine (interleukin-8 (IL-8)), found in excessive quantities in people with IBD.
Caprylic acid also plays a vital role in the body's regulation of energy input and output, a function that is executed by the hormone ghrelin also called the hunger hormone. The acylated form of ghrelin contributes to hunger, and unacylated ghrelin is believed to improve glycemic control and suppress hunger. The caprylic acid is assumed to augment the production of unacylated ghrelin, thus suppress hunger and maintain satiety (a feeling of fullness). Hence caprylic acid is the popular ingredient in weight-loss supplements.
There is also growing evidence that a ketogenic diet with caprylic acid can reduce the number of seizures or the severity of seizures in people with epilepsy.
Caprylic can also slow down the progression of mild to moderate Alzheimer's disease. Caprylic acid is also the active ingredient of a supplement called Axona, which is marketed as a "medical food" for people with Alzheimer's.
Caprylic acid is also used as an antimicrobial pesticide as a food contact surface sanitizer in commercial food handling establishments such as food processing equipment, dairy equipment, wineries, breweries, and beverage processing plants [21,22, 23,24].
Caprylic Acid Side Effects
Caprylic acid is possibly safe for most people when used at approved doses for nutritional supplementation. However, diets containing high amounts of caprylic acid can cause some side effects, including bloating, nausea, vomiting, diarrhea, constipation, stomach pain, growth problems in children, low levels of calcium in the blood, and an increased risk of brittle bones in people with osteoporosis.
People with liver disease or hypotension should avoid a high intake of caprylic acid and also be avoided in persons with medium-chain acyl-CoA dehydrogenase (MCAD) deficiency due to their inability to break down fatty acids efficiently which can trigger hypoglycemia (low blood sugar), vomiting, and, in severe cases, coma [23].
Suggested Dose
Most of the supplements contain up to 500 milligrams of caprylic acid, a much safer amount. For yeast infections and thrush, caprylic acid is typically sold as a "fungal defense" supplement in form of the capsules. The majority of the products are marketed in the form of multi-ingredient supplements instead of as a typical caprylic acid supplement [23].
To conclude,
Caprylic acid is a medium-chain fatty acid with potent antibacterial,
antifungal, antiviral and anti-inflammatory activities. Several in vitro
and in vivo animal studies demonstrated the effectiveness of caprylic
acid towards Candida yeast and other pathogens. It generally acts by the
disruption of biological membranes in the microorganisms and exhibits
very few side effects at nutritional doses. The synergistic combinations
of naturally-derived components with caprylic acid can be the promising
substitutes for synthetic compounds against various microbial
infections and other diseases.
About the Author

Dr. Harmeet Kaur received her Bachelors in Pharmacy from Guru Nanak Dev University in Amritsar, India in 2000. Guru Nanak Dev University is a state owned university with an "A" grade nationally.
Dr. Kaur received her Masters in Medicinal Chemistry from the National Institute of Pharmaceutical Education and Research in 2002.
In 2015 Dr. Kaur was awarded her Ph.D in Pharmaceutical Sciences from Maharshi Dayanad University in Rohtak, India.
Dr. Kaur is presently a Senior Research Scientist at Maharshi Dayanand University in India.
Dr. Kaur has over 35 published Research papers concerning infectious diseases caused by yeasts, fungi, and bacteria using both prescription drugs and natural plant compounds. She has also performed many studies on cancer cells.
Of particular importance to us, is her multiple experiments performed on Candida albicans and pathogenic bacteria using natural compounds. Because of this experience, we are thrilled to have her on the YeastInfectionAdvisor team.
Back to Herbal Yeast Infection Remedies
Any questions about caprylic acid or yeast infections in general, please feel free to contact us using the form on the contact page of this website or see your doctor.
Dr. Kaur's Medical References
[1].
Beare-Rogers JL, Dieffenbacher A, Holm JV. Lexicon of lipid nutrition
(IUPAC Technical Report). Pure and Applied Chemistry. 2001;73(4):685-744.
[2]. https://www.healthline.com/health/caprylic-acid-coconut-oil
[3].
Papamandjaris AA, MacDougall DE, Jones PJ. Medium chain fatty acid
metabolism and energy expenditure: obesity treatment implications. Life
sciences. 1998;62(14):1203-1215.
[4].
Yoon BK, Jackman JA, Valle-González ER, Cho NJ. Antibacterial free
fatty acids and monoglycerides: biological activities, experimental
testing, and therapeutic applications. International journal of
molecular sciences. 2018;19(4):1114.
[5].
Batovska DI, Todorova T, Tsvetkova V, Najdenski HM. Antibacterial study
of the medium chain fatty acids and their 1-monoglycerides: individual
effects and synergistic relationships. Polish Journal of Microbiology. 2009;58(1):43-47.
[6]. Marounek M., Skřivanová E., Rada V. Susceptibility of Escherichia coli to C2–C18 fatty acids. Folia Microbiologica. 2003;48:731–735.
[7]. Skřivanová E, Savka OG, Marounek M. In vitro effect of C 2–C 18 fatty acids on salmonellas. Folia microbiologica. 2004;49(2):199.
[8].
Kollanoor A, Vasudevan P, Nair MK, Hoagland T, Venkitanarayanan K.
Inactivation of bacterial fish pathogens by medium‐chain lipid molecules
(caprylic acid, monocaprylin and sodium caprylate). Aquaculture
Research. 2007;38(12):1293-1300.
[9].
Nair MK, Joy J, Vasudevan P, Hinckley L, Hoagland TA, Venkitanarayanan
KS. Antibacterial effect of caprylic acid and monocaprylin on major
bacterial mastitis pathogens. Journal of dairy science. 2005;88(10):3488-3495.
[10].
Díaz De Rienzo MA, Stevenson P, Marchant R, Banat IM. Antibacterial
properties of biosurfactants against selected Gram-positive and-negative
bacteria. FEMS Microbiology Letters. 2016;363(2):224.
[11].
Valipe SR. Investigating the antimicrobial effect of caprylic acid and
its derivatives on Dermatophilus congolensis and developing a species
specific PCR to detect Dermatophilus congolensis. University of
Connecticut; 2011.
[12].
Elmore LK, Nance G, Singleton S, Lorenz L. treatment of dermal
infections with topical coconut oil. Natural Medicinal Journal. 2014;6(5):1-3.
[13].
Marounek M, Putthana V, Benada O, Lukešová D. Antimicrobial activities
of medium-chain fatty acids and monoacylglycerols on Cronobacter
sakazakii DBM 3157T and Cronobacter malonaticus DBM. Czech Journal of
Food Sciences. 2012;30(6):573-580.
[14].
Kim SA, Rhee MS. Synergistic antimicrobial activity of caprylic acid in
combination with citric acid against both Escherichia coli O157: H7 and
indigenous microflora in carrot juice. Food microbiology. 2015;49:166-172.
[15]. Tsukahara T. Fungicidal action of caprylic acid for Candida albicans. Japanese Journal of Microbiology. 1961;5(4):383-94.
[16].
Neuhauser I. Successful treatment of intestinal moniliasis with fatty
acid-resin complex. AMA Archives of Internal Medicine. 1954;93(1):53-60.
[17].
Bergsson G, Arnfinnsson J, Steingrı́msson Ó, Thormar H. In vitro
killing of Candida albicans by fatty acids and monoglycerides.
Antimicrobial agents and chemotherapy. 2001;45(11):3209-3212.
[18].
Bae YS, Rhee MS. Short-term antifungal treatments of caprylic acid with
carvacrol or thymol induce synergistic 6-log reduction of pathogenic
candida albicans by cell membrane disruption and efflux pump inhibition.
Cellular Physiology And Biochemistry. 2019;53(2):285-300.
[19].
Rosenblatt J, Reitzel RA, Raad I. Caprylic acid and glyceryl trinitrate
combination for eradication of biofilm. Antimicrobial Agents and
Chemotherapy. 2015;59(3):1786-8.
[20].
Rosenblatt J, Reitzel RA, Vargas-Cruz N, Chaftari AM, Hachem R, Raad I.
Caprylic and polygalacturonic acid combinations for eradication of
microbial organisms embedded in biofilm. Frontiers in Microbiology. 2017;8:1999.
[21].
Franzosa EA, Sirota-Madi A, Avila-Pacheco J, Fornelos N, Haiser HJ,
Reinker S, Vatanen T, Hall AB, Mallick H, McIver LJ, Sauk JS. Gut
microbiome structure and metabolic activity in inflammatory bowel
disease. Nature Microbiology. 2019;4(2):293-305.
[22].
Lemarié F, Beauchamp E, Legrand P, Rioux V. Revisiting the metabolism
and physiological functions of caprylic acid (C8: 0) with special focus
on ghrelin octanoylation. Biochimie. 2016;120:40-48.
[23]. https://www.webmd.com/vitamins/ai/ingredientmono-1465/caprylic-acid
[24].
Costa AR, Rosado EL, Soares-Mota M. Influence of the dietary intake of
medium-chain triglycerides on body composition, energy expenditure and
satiety; a systematic review. Nutricion Hospitalaria. 2012;27(1):103-8.
Home Privacy Policy Copyright Policy Disclosure Policy Doctors Store
Copyright © 2003 - 2025. All Rights Reserved under USC Title 17. Do not copy
content from the pages of this website without our expressed written consent.
To do so is Plagiarism, Not Fair Use, is Illegal, and a violation of the
The Digital Millennium Copyright Act of 1998.