- Home
- Herbal Remedies
- Thyme Leaf
Thyme Leaf for Yeast & Bacterial Infections
Updated 2/27/2021
Written By Pharmaceutical Scientist Dr. Harmeet Kaur, PhD
This article talks about Thyme Leaf's effects on yeasts, bacteria, its other uses and health benefits, and possible safety issues and side effects.
Thyme leaf or Thymus vulgaris L. belongs to the category of aromatic plants and representative of the family Lamiaceae has a long history of use in both folk and conventional medicine. It is mainly indigenous to Europe, specifically the Mediterranean region, and is a small, bushy herb, with small, elliptical, greenish-grey, and shortly-stalked leaves. Thyme has a characteristic odor of thymol and is used as a culinary herb.
In native medicine, leaves and flowering parts of Thymus species have been extensively used as a tonic, herbal tea, carminative, antiseptic, and antitussive, as well as for treating colds. Thyme essential oil (TEO) generally obtained by distillation or cold pressing method from leaves and flowers has a pungent odor due to the presence of more than 44% phenols, mainly thymol, and carvacrol and have antimicrobial properties. As a result of anti-bacterial properties, it is used as a disinfectant and main active ingredient in toothpaste and Listerine [1,2].
Chemical Constituents Present in Thyme Leaf
The active principle present in the leaves of various species of thyme is an essential oil (Thymi aetheroleum) which is only 0.8 to 2.6% with 45-47% thymol, 32-34% p-cymene, 10-15% γ-terpinene, 4-5% carvacrol as dominant compounds. The other components present in essential oil are oxygenated monoterpenes such as 1,8 cineole (0.8-2.17%), linalool (2.03-6.8%), menthone (2.2%), neomenthol (2.8%), borneol (2.6-8.9%), terpinen4-ol (1-2.63%); Sesquiterpene hydrocarbons like trans-caryophyllene (1.13–5.1%), β-cubebene (2.4%); Oxygenated sesquiterpenes e.g caryophyllene oxide (0.33–4.6 %); aliphatic compounds such as 1-octen-3-ol (2.8%).
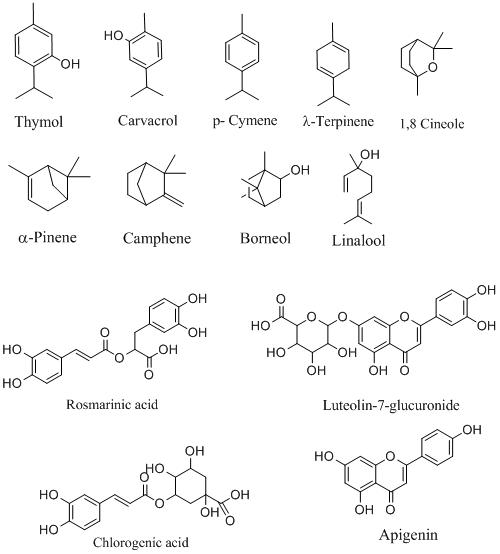
The percentage of the main constituents varies significantly, depending on endogenous and exogenous factors such are: ontogenesis, chemotype, climatic and geographic conditions, methods intended for processing of plant material, and essential oil isolation. However, aromatic oxygenated monoterpenes should constitute a minimum of 40% of the thyme essential oil in each case.
Thyme leaves are also found to be a rich source of several phenolic compounds, mainly flavonoids, phenolic acids, and other secondary metabolites such as caffeic acid, chlorogenic acid, luteolin, luteoline-7-O-glucuronide, rosmarinic acid, hispidulin, apigenin, oleanolic acid, ursolic acid, arbutin, hydroquinone, naringenin, naringenin-7-O-ß-glucoside, narirutin, eriodictyol, eriodictyol-7-O-ß-glucoside, eriocitrin that are anticipated to contribute to biological activities of thyme.
Leaves are also a good source of vitamins particularly rich in Vitamin A and Vitamin C. Vitamin A is vital for maintaining healthy mucus membranes and skin along with good vision. Vitamin C has antioxidant properties and helps the body to fight against infectious diseases and destroy the harmful pro-inflammatory free radicals. Leaves also comprise of Vitamin B6 (pyridoxine), which assists in maintaining GABA levels in the brain, and acts as a stress buster.
Vitamin E, Vitamin K, and folic acid are other vitamins found in this herb. Thyme leaves are also packed with minerals like potassium, iron, calcium, manganese, selenium, and magnesium. Potassium, being a vital component of body fluids and cells, controls blood pressure and heart rate. Iron is intricated in the formation of red blood cells.
Manganese act as a co-factor for the antioxidant enzyme superoxide dismutase. Thyme essential oil, carvacrol, and thymol are generally recognized as safe (GRAS status) and have been registered by the European Commission for use as flavoring agents in foods [3,4].
Thyme Leaf's Anti-bacterial Activity
Recently, several studies have confirmed the potential of thyme essential oil and isolated dominant monoterpenes as antibacterial and antifungal agents, together with the inhibition of mycotoxin production.
Thyme proves to be an effective antimicrobial agent for Salmonella typhimurium, S. enteritidis, Escherichia coli, Y. enterocolitica, Listeria monocytogenes, Shigella sonnei, S. flexneri, and Staphylococcus aureus. Neda Gavarić et al. evaluated the antibacterial potential of the extracts attained from the waste material left after the preparation of drug from stem and isolation of thyme essential oil (deodorized leaves) by the broth dilution method towards various clinical isolates of bacterial strains (S. aureus, S. infantis, Bacillus cereus, E. coli, and Campylobacter jejuni).
The HPLC analysis has shown that the extracts, especially attained from deodorized leaves (after removal of essential oil by hydrodistillation) represent a valuable source of luteolin 7-O-glucuronide and rosmarinic acid. The extracts possessed strong antimicrobial activity than standard thyme leaf extract. No cytotoxicity nor cytoprotection was determined. Moreover, all investigated thyme extracts exhibited lower MIC values on all investigated bacterial strains in comparison to isolated rosmarinic acid. This indicated that phenolic compounds possess a synergistic antibacterial effect, which is important in terms of the emerging occurrence of resistance [5].
In a study by Shahad Alsaraf et al, Thyme essential oil (TEO) isolated from an Omani cultivar was examined for its chemical composition, antioxidant, and antimicrobial activity. A total of 11 volatile constituents representing 99.71% of the total detected components were identified in TEO by GC-MS analysis. TEO exhibited moderate in vitro free radical scavenging activity in the DPPH assay method. However, TEO exhibited potent antibacterial activity against both S. aureus and E. coli and is categorized as bactericidal.
The antioxidant and antimicrobial activity of TEO is credited to its chemical composition and mainly attributable to its major chemical constituent carvacrol. In silico prediction and molecular docking studies of its major constituent, carvacrol also supported the experimental results. Interestingly, TEO demonstrated a better antibacterial spectrum in comparison to standard drug Ampicillin against both the strains even at a low concentration of 5 μL [6].
A study by Khan et al. also evaluated the antibacterial activity of the Pakistani variety of TEO at 15 μL and obtained the zone of inhibitions (30 mm and 25 mm) against E. coli and S. aureus [7]. Yamazaki et al., have also supported the same results by correlating the antibacterial results to the presence of phenolic compounds such as thymol and carvacrol. They found thyme oil to exhibit the greatest antibacterial activity against E. coli O157:H7 [8].
Al-Saimary et al assessed the antibacterial activity of various concentrations of alcoholic and aqueous extracts of thymus leaves and flowers against clinical isolates of E. coli, Enterobacter sp., Klebsiella sp., Proteus sp., Pseudomonas aeroginosa, S. aureus, Staph. epidermidis, Streptococcus faecalis, and B. subtilis by agar diffusion and tube dilution techniques. Alcoholic extracts were found to be more effective in comparison to aqueous extracts and found to be more efficacious on Gram-positive bacteria than on Gram-negative bacteria. The biggest inhibition zone was recorded against S. faecalis at 1mg/mL of mixed alcoholic extracts was (35 mm). The MICs of all extracts were found in the range of (400-1300) µg/mL [9].
Zeinab Mohsenipour evaluated the antimicrobial activity of T. vulgaris extracts from leaves against planktonic form and biofilm structures of six pathogenic bacteria by disc diffusion technique. These bacteria were S. aureus, B. cereus, Streptococcus pneumoniae, P. aeruginosa, E. coli, and Klebsiella pneumonia. T. vulgaris extracts had shown a high ability to inhibit the growth of P. aeruginosa and S. aureus.
However, these extracts had shown low inhibition efficiency against B. cereus and E. coli and did not show an inhibitory effect on other tested bacterial pathogens. Interestingly, T. vulgaris extracts inhibited the growth of all tested bacteria in low concentration in broth media than on solid media. With these results, it can be concluded that similar to other plant extracts antimicrobial constituents in T. vulgaris extracts have low diffusion in solid media compared with broth media. Hence, for a favorable impact on solid media, a much higher concentration than broth media is needed [10].
Mohammadmehdi Fani et al demonstrated the strong in vitro antimicrobial activity of Thymus vulgaris oil on clinical isolates of major oral pathogens such as S. mutans, S. pyogenes, C albicans, A actinomycetemcomitans, and P gingivalis and hence might be used in toothpaste, mouth rinse, or aromatherapy for prevention and treatment of associated oral infections [11].
How Thyme Leaf Works Against Bacteria
The antibacterial activity of Thyme extracts may be due to the presence of phenolic constituents (thymol and carvacrol), which make up a large percentage of the volatile oil. Thymol is 25 times more effective in comparison to phenol with less toxicity and exerts its antimicrobial action via binding to membrane proteins by hydrogen and hydrophobic bonding resulted in alteration in the permeability of the membranes.
Thymol also diminished intracellular adenosine triphosphate (ATP) content of E. coli and enhanced extracellular ATP, resulted in disruption of the functions of plasma membranes.
Mostly extracts, in general, thyme extracts are more effective on Gram-positive bacteria than on Gram-negative bacteria, maybe due to the nature of bacterial cell membranes. The cell membrane of Gram-positive bacteria comprises of mucopolysaccharides, proteins, and fewer amounts of phospholipids, while Gram-negative bacteria have huge amounts of phospholipids and more pores in the cell envelope.
Thus, the permeability, entry, and reaction of most antibiotics and/or antimicrobial agents through cell envelope (the outer and cytoplasmic membrane) are highly efficient for Gram-positive bacteria depending on the reaction with the protein layer (peptidoglycans or mucopolysaccharide) [12].
Thyme Leaf's Anti-Yeast Activity
Deaths related to fungal infections are mostly associated with Cryptococcus (that causes cryptococcosis, an infection commonly associated with immunosuppressive individuals), Candida (Candidiasis such as oral and vulvovaginal), Aspergillus (Aspergillosis), and Pneumocystis (pneumonia). Thyme essential oils are speciously among the greatest inhibitors of fungal microorganisms because of the presence of the terpenic phenolic compounds such as thymol, carvacrol as main components. Thyme oils have been used in folk medicine in different communities for patients suffering from mycotic infections.
Eugénia Pinto et al find the composition of the essential oil from Thymus pulegioides aerial parts isolated via hydrodistillation and evaluated its antifungal activity on various fungal strains such as Candida strains (C. albicans (M1, H37), C. krusei (H9), C. tropicalis (H18), C. glabrata (H16, H30), C. guillermondii (Mat23), isolated from recurrent cases of vulvovaginal candidiasis); Aspergillus strains (A. niger (F01), A fumigatus (F05, F07, and F17) and A. flavus (F44) isolated from bronchial secretions, and dermatophyte strains (Microsporum canis FF1, M. gypseum FF3, Trichophyton mentagrophytes FF7, T. rubrum FF5, and Epidermophyton floccosum FF9) isolated from nails and skin. The oil showed high contents of thymol and carvacrol. The MIC values of oil were ranged from 0.16 to 0.32 µl/mL against dermatophyte and Aspergillus strains.
The oil showed the highest MIC values, ranging from 0.32 to 0.64 µl/mL against Candida. This essential oil found to a broad-spectrum agent that inhibits not only Aspergillus, dermatophytes, and Candida species, but also fluconazole-resistant C. albicans isolates, and C. glabrata and C. krusei, which are intrinsically resistant to fluconazole or whose resistance is easily inducible. The mechanism of action by which oil act mainly due to lesion formation in the cytoplasmic membrane and disruption of normal sterol biosynthetic pathways, leading to a reduction in ergosterol biosynthesis [13].
M. Moghtader et al study the antifungal effects of the oil from Thymus vulgaris against A. niger by disc diffusion method. Thirty-two compounds have been identified in the essential oil of this plant with Thymol (32.67%), P-cymene (16.68%), γ-terpinene (12.65%), and Carvacrol (8.32%) as the main components. The results revealed that essential oil from thymus plant at 1, 1/2, and 1/4 oil dilutions exhibit strong antifungal activity in comparison to standard antibiotics like Gentamycin (8 mg/mL) and Streptomycin sulfate (72% SP). The synthetic thymol exhibited moderate activity at 1% dilution but exhibited strong activity at 10% dilution [14].
In another study, Melissa Alves et al evaluated the antifungal effect of the essential oils of two thyme species, Thymus camphoratus and Thymus carnosus, used in traditional medicine in Portugal. The antifungal potential of the essential oils was evaluated against both clinically isolated and reference strains of various filamentous fungi and yeast. Yeast strains included C. albicans, C. guilliermondii, C. krusei, C. parapsilosis, C. tropicalis, and Cryptococcus neoformans; Dermatophyte strains included Epidermophyton floccosum, Microsporum canis, M. gypseum, Trichophyton mentagrophytes, T. mentagrophytes var. interdigitale, T. rubrum and T. verrucosum whereas Aspergillus strains were A. flavus, A. fumigatus, and A. niger.
A special focus was placed on their effect on C. albicans virulence factors since the yeast-to-hypha transition represents the main virulence factor associated with candidiasis. The essential oils were also assessed for their safety profile on keratinocytes. The effect on C. albicans germ tube formation, metabolism, and biofilm disruption were also considered. T. camphoratus oil was found to rich in 1,8-cineole and α-pinene whereas T. carnosus oil showed high amounts of borneol and camphene as shown by GCMS analysis.
The essential oils of both species demonstrated a slight fungicidal effect for most of the strains of Candida spp. The inhibition of germ tube formation in C. albicans has been due to linalool, a minor component present in both oils. Stimulatingly, although phenolic thyme essential oils, such as those from T. zygis and T. vulgaris, show potent antifungal activities but very less effective in inhibition of C. albicans germ tube formation in comparison to T. camphoratus and T. carnosus oils. Both oils showed a fungicidal effect against dermatophytes, but T. camphoratus was more effective than T. carnosus, presenting lower MIC values for most of the tested strains.
Both oils were found to more active against Cryptococcus neoformans and dermatophytes and very effective in inhibiting C. albicans germ tube formation, at doses well below their MIC values and to a higher extent in comparison to the isolated compounds and fluconazole, well known antifungal drug. The oils also disrupted preformed C. albicans biofilms. Additionally, no toxicity was detected at pharmacologically relevant concentrations towards keratinocytes [15].
How Thyme Leaf Works Against Yeast & Fungi
The antifungal activity is probably because of the presence of terpenic phenols such as thymol, carvacrol, etc. which are lipophilic and can act in fatty acyl chains of membrane lipid bilayers, and alters the fluidity and permeability of cell membranes resulting in leakage of the cytoplasmic contents and cell death. Other mechanisms also have been hypothesized such as damage of membrane and cell wall with disruption linked to morphological deformation, deterioration, collapse, and of the conidia and/or hyphae.
Carvacrol and thymol have a stronger antifungal capacity, indicating more susceptibility of Aspergillus spp. than that of Penicillium spp. Thyme essential oil showed its capabilities of inhibiting fungal development causing leakage of the cytoplasm of A. flavus and was responsible for degenerative alterations in hyphae alterations, which appeared degraded or with the complete absence of conidia. Some reports have also shown the capabilities of inhibiting aflatoxin production [16,17].
Thyme Leaf's Other Uses
The thyme, its leafy parts, and oil have been used for many centuries as a culinary herb, in foods for flavor, aroma, and preservation, and as herbal medicine.
The plant is useful as an infusion to treat diabetes, cough, and cold and chest infections, bronchitis, whooping cough, and as a syrup for digestive upset, stomach pain (gastritis), intestinal gas (flatulence), diarrhea, a movement disorder in children (dyspraxia) and bedwetting.
It is also used for soothing sore throat, as thyme has antiseptic, antibacterial, and antifungal properties.
Thyme has also been used as anthelmintic, astringent, carminative, tonic, and disinfectant.
Thyme oil is used as a germ-killer in liniments and mouthwashes. It has also applications in the treatment of baldness and ear infections.
The plant has been reported incredibly useful in cases of assorted intestinal infections and infestations, such as hookworms, ascariasis, etc.
Thyme also has spasmolytic effects as flavonoids in thyme can relax smooth muscles of ileum by blocking the acetylcholine and histamine receptors, and/or antagonism of the calcium channels.
Additional activities related to thyme include treatment of cartilaginous tube, bronchial, and urinary infections; appetite stimulant effect; liver function improvement; and treatment of laryngitis and inflammation.
Thyme and thyme oil have also demonstrated strong antioxidant properties.
Furthermore, the contained flavonoids in thyme prevent the release of superoxide anion and protect the erythrocytes from oxidative stress.
Thymol and carvacrol exerted anti-inflammatory effects by inhibiting the synthesis of prostaglandins.
Thyme is also used for skin issues like oily skin, acne, sciatica, dermatitis, relieves neuralgia, and relieves bites and stings.
The essential oil can be used as a rub for rheumatic pain or aching joints, and also be used in the treatment of athlete’s foot (Tinea pedis) [18, 19, 20].
Thyme Leaf's Side Effects
Thyme is considered safe when taken by mouth as medicine in a prescribed dose for short periods.
In some people, it can cause headaches, dizziness, asthma, muscle weakness, allergic reactions ranging from contact dermatitis to hay fever symptoms, conjunctivitis, and digestive system upset.
It should be avoided in case of allergy or hypersensitivity to thyme.
The oral ingestion or non-diluted topical application of thyme oil should be evaded due to potential toxicity.
Also, topical preparations in the areas of skin injury or atopic patients due to multiple reports of contact dermatitis may not be applied.
Thyme should be used with caution in persons with thyroid disorders due to reported anti-thyrotropic effects in animal research of the allied species Thymus serpyllum.
It should not be consumed in pregnancy or breastfeeding [21].
Thyme Leaf Conclusion
Thymus vulgaris is a vital aromatic and medicinal plant which has a long history of traditional use as food preservatives, aromatic ingredient and in phytopharmaceutical preparations. Thyme essential oil contains several bioactive components such as terpenes like thymol, carvacrol, and linalool, having anti-inflammatory, antioxidative, antibacterial, and antifungal properties.
About the Author

Dr. Harmeet Kaur received her
Bachelors in Pharmacy from Guru Nanak Dev University in Amritsar, India
in 2000. Guru Nanak Dev University is a state owned university with an
"A" grade nationally.
Dr. Kaur received her Masters in Medicinal
Chemistry from the National Institute of Pharmaceutical Education and
Research in 2002.
In 2015 Dr. Kaur was awarded her Ph.D in Pharmaceutical Sciences from Maharshi Dayanad University in Rohtak, India.
Dr. Kaur is presently a Senior Research Scientist at Maharshi Dayanand University in India.
Dr.
Kaur has over 35 published Research papers concerning infectious
diseases caused by yeasts, fungi, and bacteria using both prescription
drugs and natural plant compounds. She has also performed many studies
on cancer cells.
Of particular importance to us, is her multiple experiments performed on Candida albicans
and pathogenic bacteria using natural compounds. Because of this experience, we are thrilled to have her on the YeastInfectionAdvisor team.
Back to Herbal Yeast Infection Remedies
If you have any questions about thyme leaf or yeast and bacteria infections in general, please feel free to contact us from the Contact page of this website or talk to your doctor.
Dr. Kaur's Medical References
[1]. Dauqan EM, Abdullah A. Medicinal and functional values of thyme (Thymus vulgaris L.) herb. Journal of Applied Biology & Biotechnology. 2017;5(2):17-22.
[2]. Prasanth Reddy V, Ravi Vital K, Varsha PV, Satyam S. Review on Thymus vulgaris traditional uses and pharmacological properties. Medicinal & Aromatic Plants. 2014;3(164):2167-0412.
[3]. Al Maqtari MA, Alghalibi SM, Alhamzy EH. Chemical composition and antimicrobial activity of essential oil of Thymus vulgaris from Yemen. Turkish Journal of Biochemistry. 2011;36:342-9.
[4] Azaz AD, Irtem HA, Kurkcuoǧlu M, Baser KH. Composition and the in vitro antimicrobial activities of the essential oils of some Thymus species. Zeitschrift für Naturforschung C. 2004;59(1-2):75-80.
[5]. Gavarić N, Kovač J, Kretschmer N, Kladar N, Možina SS, Bucar F, Bauer R, Božin B. Natural products as antibacterial agents—Antibacterial potential and safety of post-distillation and waste material from Thymus vulgaris L., Lamiaceae. Concepts, Immunology, and Microbiology: Compounds and the Alternatives of Antibacterials. Rijeka, Croatia: InTech. 2015 Dec 9:123-52.
[6]. Alsaraf S, Hadi Z, Al-Lawati WM, Al Lawati AA, Khan SA. Chemical composition, in vitro antibacterial and antioxidant potential of Omani Thyme essential oil along with in silico studies of its major constituent. Journal of King Saud University-Science. 2020;32(1):1021-8.
[7]. Inayatullah S, Shafee M, Shafiq M, Asif M, Parveen S, Kakar K, Khan YH, Amir M, Qadir A, Yakoob M, Baloch A. Antimicrobial activity of Thyme (Thymus vulgaris) essential oil cultivated in Quetta, Balochistan, Pakistan. International Journal of Biosciences. 2017;10:105-10.
[8]. Yamazaki K, Yamamoto T, Kawai Y, Inoue N. Enhancement of antilisterial activity of essential oil constituents by nisin and diglycerol fatty acid ester. Food Microbiology. 2004;21(3):283-9.
[9]. Al-Saimary IE, Bakr SS, Khudaier BY, Abass YK. Efficiency of antibacterial agents extracted from Thymus vulgaris L.(Lamiaceae), part 2. The Internet Journal of Wellness. 2006;4(1):1-5.
[10]. Mohsenipour Z, Hassanshahian M. The inhibitory effect of Thymus vulgaris extracts on the planktonic form and biofilm structures of six human pathogenic bacteria. Avicenna journal of phytomedicine. 2015;5(4):309.
[11]. Fani M, Kohanteb J. In vitro antimicrobial activity of Thymus vulgaris essential oil against major oral pathogens. Journal of evidence-based complementary & alternative medicine. 2017;22(4):660-666.
[12]. Xu J, Zhou F, Ji BP, Pei RS, Xu N. The antibacterial mechanism of carvacrol and thymol against Escherichia coli. Letters in applied microbiology. 2008;47(3):174-179.
[13]. Pinto E, Pina-Vaz C, Salgueiro L, Gonçalves MJ, Costa-de-Oliveira S, Cavaleiro C, Palmeira A, Rodrigues A, Martinez-de-Oliveira J. Antifungal activity of the essential oil of Thymus pulegioides on Candida, Aspergillus and dermatophyte species. Journal of medical microbiology. 2006;55(10):1367-73.
[14]. Moghtader M. Antifungal effects of the essential oil from Thymus vulgaris L. and comparison with synthetic thymol on Aspergillus niger. Journal of Yeast and Fungal Research. 2012;3(6):83-88.
[15]. Alves M, Gonçalves MJ, Zuzarte M, Alves-Silva JM, Cavaleiro C, Cruz MT, Salgueiro L. Unveiling the antifungal potential of two Iberian thyme essential oils: effect on C. albicans germ tube and preformed biofilms. Frontiers in pharmacology. 2019;10:446.
[16]. Romo JA, Pierce CG, Chaturvedi AK, Lazzell AL, McHardy SF, Saville SP, Lopez-Ribot JL. Development of anti-virulence approaches for candidiasis via a novel series of small-molecule inhibitors of Candida albicans filamentation. MBio. 2017;8(6).
[17]. Richardson M, Lass‐Flörl C. Changing epidemiology of systemic fungal infections. Clinical Microbiology and Infection. 2008;14:5-24.
[18]. Almanea A, Abd El-Aziz GS, Ahmed MM. The Potential Gastrointestinal Health Benefits of Thymus Vulgaris Essential Oil: A Review. Biomedical and Pharmacology Journal. 2019;12(04):1793-9.
[19]. Hosseinzadeh S, Kukhdan AJ, Hosseini A, Armand R. The application of Thymus vulgaris in traditional and modern medicine: a review. Global J Pharmacol. 2015;9:260-266.
[20]. Goudjil MB, Zighmi S, Hamada D, Mahcene Z, Bencheikh SE, Ladjel S. Biological activities of essential oils extracted from Thymus capitatus (Lamiaceae). South African Journal of Botany. 2020;128:274-282.
[21]. https://www.rxlist.com/thyme/supplements.htm
Home Privacy Policy Copyright Policy Disclosure Policy Doctors Store
Copyright © 2003 - 2025. All Rights Reserved under USC Title 17. Do not copy
content from the pages of this website without our expressed written consent.
To do so is Plagiarism, Not Fair Use, is Illegal, and a violation of the
The Digital Millennium Copyright Act of 1998.