- Home
- Herbal Remedies
- Olive Leaf
Olive Leaf as an Antimicrobial Agent
Updated 11/02/2021
Written By Pharmaceutical Scientist Dr. Harmeet Kaur, PhD
Olive leaf effects against bacteria, yeasts, viruses, other health benefits and possible side effects are all discussed below.
Olive leaf (Olea europaea L.) is one of the most important trees whose fruit, leaves, and oil have an ancient history of traditional, nutritional, and medicinal usages. Olive is the only species from the genus Olea which constitutes a vital part of the Mediterranean diet, the highest value of which may be due to olive polyphenols that modulate part of the oxidative balance in vivo.
Historically, olive leaves have been widely used as a remedy for the treatment of diseases like malaria and fever in European and other Mediterranean countries. The polyphenols present in olive leaves become a subject of intensive investigation due to their various beneficial effects like antioxidant, anticarcinogenic, anti-diabetic, anti-atherosclerotic, anti-hypertensive, antimicrobial, and anti-inflammatory activities.
Qualities of olive leaf extract are mostly correlated to the content of the low molecular weight polyphenol oleuropein (up to 60-90 mg/g dry leaves weight), as well as present flavonoids. Moreover, the different phenolic compounds present in the extract have expressively higher antimicrobial activity in comparison to the individual phenolics [1, 2].
Chemical Constituents Present in Olive Leaf
The main biochemicals present in the olive tree are secoiridoids, terpenoids, flavonoids carbohydrates, sugar alcohols, etc. The olive leaves mainly comprise of the secoiridoids such as oleuropein, methyloleuropein, ligstroside, and oleoside; flavonoids such as kaempferol, apigenin -7-O-rutinoside, luteolin-7-O-glucoside, and chrysoeriol; and phenolic compounds like caffeic acid, tyrosol, and hydroxytyrosol.
Oleuropein is one of the most abundant bioactive components contained in the olive leaves. Oleuropein which is mainly a phenolic glycoside comprises a polyphenol known as hydroxytyrosol (4-(2-hydroxyethyl) benzene-1,2-diol), a secoiridoid called elenolic acid, and a glucose molecule.
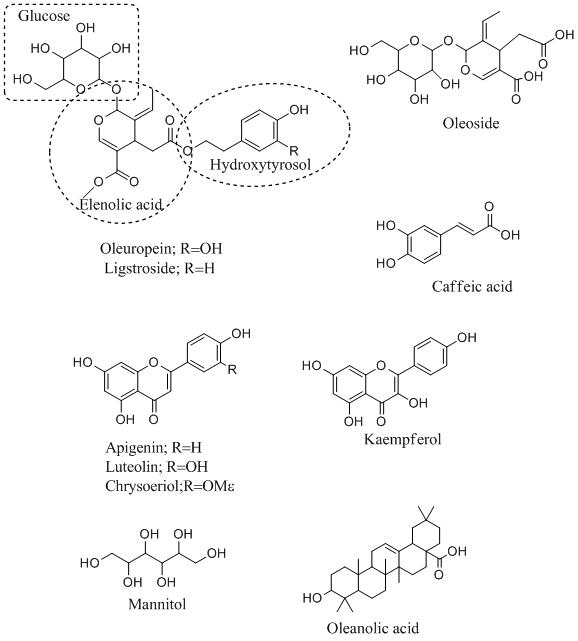
Secoiridoids, generally produced by secondary metabolisms of terpenes as the pioneers of various indole alkaloids. The content of oleuropein varies depending on the variety, climate, organ, olive product, ripeness of the olives at harvesting, and the processing system employed. Upon hydrolysis, oleuropein can produce elenolic acid, hydroxytyrosol, tyrosol, and glucose.
Oleuropein and hydrolysis products found in olive leaf have important biological characteristics. It is considered that hydroxytyrosol is particularly correlated with the health benefits of olive products.
Mannitol, another component present in olive leaf, consists of 3% of the dry weight of olive leaf, and due to its sweetness ratio and low energy content, it is used as an additive in the drug and food industry. The oleanolic acid is the main triterpene present in the olive leaf comprised of 3% of the leaf’s dry weight and has very important pharmacological features [3,4].
Antimicrobial Activity of Olive Leaf
Olive leaf compounds such as oleuropein and its derivatives including tyrosol and hydroxytyrosol exhibit antimicrobial activities, which can reduce the risk of microbial infections. These compounds can inhibit bacteria, viruses, retroviruses, fungi, yeasts, mycoplasmas, molds, and other parasites, mainly in the gastrointestinal and respiratory tracts.
Does Olive Leaf Have Antibacterial Activity?
The evaluation of phenolic compounds present in olive leaves aqueous extract has been done for their in vitro activity against several microorganisms, namely Gram-positive (Bacillus cereus, B. subtilis, and Staphylococcus aureus), Gram-negative bacteria (Escherichia coli, Pseudomonas aeruginosa, and Klebsiella pneumoniae), and fungi (Cryptococcus neoformans and Candida albicans). Apart from the concerns of food quality degradation, these microorganisms also are the causal agents of the respiratory tract and intestinal infections of humans. The olive leaves extract repressed all the tested bacteria and fungi, signifying a broad spectrum of antimicrobial activity in a concentration-dependent manner. B. cereus (Gram-positive) and C. albicans (fungi) both presenting IC25 values lower than 1 mg/mL were found to be the most sensitive microorganisms while B. subtilis was the most resistant microorganism [5].
Yanhong Liu et al investigated the antimicrobial effect of olive leaf extract against major foodborne pathogens, including, E. coli O157:H7, Listeria monocytogenes, and Salmonella enteritidis. The results have shown that olive leaf extract at a concentration of 62.5 mg/mL, completely inhibited the growth of all three pathogens. Also, the extract has reduced the cell motility in L. monocytogenes, correlated to the absence of flagella as shown by scanning electron microscopy. Furthermore, the extract has also inhibited biofilm formation in S. enteritidis and L. monocytogenes [6].
Sudjana et al investigated the activity of a commercial extract derived from the olive leaves against several microorganisms (n = 122) using broth microdilution and agar dilution techniques. The extract was found to be most active against Helicobacter pylori, Campylobacter jejuni, and S. aureus [including methicillin-resistant S. aureus (MRSA)], with MICs as low as 0.31-0.78% (v/v). For all other test organisms, the extract demonstrated little activity with MICs ranging from 6.25% to 50% (v/v). Hence, the olive leaf extract can regulate the composition of the gastric flora by selectively decreasing the levels of C. jejuni and H. pylori.
M.G. Soni introduced hydroxytyrosol as an effective polyphenol in olive leaf, with toxic effects against bacteria including Pseudomonas syringae (Gram-negative) and Corynebacterium michiganense (Gram-positive). Oleuropein and hydrolysis products can inhibit the development of S. enteritidis, production and development of enterotoxin B by S. aureus, and the germination and consequent development of spores of B. cereus. Oleuropein and other phenolic compounds (vanillic acid, p-hydroxybenzoic, and p-coumaric acids) can completely inhibit the development of K. pneumoniae, B. cereus, and E. coli [7,8].
Olive Leaf's Mechanism of Antibacterial Action
The oleuropein and other similar phenolic compounds are generally classified as surface-active agents due to their ability to denature proteins. They exhibit their antibacterial effect by causing leakage of cytoplasmic constituents such as protein, phosphate, potassium, and glutamate from bacteria due to the disruption of cell peptidoglycan or damage of the cell membrane.
The mode of action of oleuropein and other polyphenolics is concentration-dependent. The higher concentration of them precipitates proteins while lower concentrations affect the activity of enzymes related to energy production. However, oleuropein proved to be expressively more toxic for Gram-positive bacteria in comparison to Gram-negative bacteria, probably due to the reduced drug capability because of the glycosidic group to penetrate the outer membrane or to reach the target site.
The antibacterial action of olive leaf polyphenolics in L. monocytogenes has been related to the cell depolarization, lower intracellular ATP, decrease in bacterial protein (reduction in protein synthesis and protein loss due to the increase in membrane permeability), and DNA (due to inhibition of DNA synthesis or stimulation of the cleavage of DNA), and cell fluid leakage accordingly of the severe destruction of the cell morphology [9].
Does Olive Leaf Have an Effect on Yeasts & Other Fungi?
Fusarium spp., Aspergillus spp., Rhizopus spp. and Penicillium spp. have been stated as the pivotal agents of foodborne diseases and food spoilage, a major problem in the world, even in well-developed countries. Antimicrobial activities of the methanol, ethyl acetate, and acetone extracts of olive leaves and some phenolic components were investigated to screen against 30 fungal strains (Aspergillus chevalieri, A. elegans, A. chrysogenum, A. flavus [three strains], A. nidulans, A. fumigatus, A. niger [two strains], A. parasiticus [four strains], A. oryzae, A. tamari, A. versicolor, A. wentii, Alternaria alternata, P. verrucosum, Fusarium oxysporum, F. semitectum, Neurospora crassa, Mucor racemosus, Rhizopus oligosporus, Penicillium citrinum, P. echinulatum, P. italicum, P. roqueforti, and P. griseofulvum,) by the disc diffusion method.
The aqueous extract was found to be the best as it completely inhibited the growth of 10 molds, followed by methanol and acetone extracts, which were effective against eight molds. The inhibition zones ranged from 7 to 21 mm. A. parasiticus was found to be the most resistant strain while A. wentii was the most sensitive on comparison of the sensitivity of the fungi with all crude olive leaf extracts and pure phenolic compounds [10].
C. albicans, the most common yeast found on the mucosal membranes of humans, can adhere to epithelial tissues and cause superficial infections such as candidiasis, the major causes of morbidity and mortality, especially in immunocompromised individuals worldwide. According to a study by Markin et al., olive leaf aqueous extracts can destroy 15% of the C. albicans within 24 h. In another study, the olive leaf aqueous extracts exhibited in vitro antifungal effects against a standard C. albicans strain (PTCC-5027) with a MIC of 24 mg/mL, MFC of 48 mg/mL, and inhibition zone diameter of 21 mm [11,12].
Korukluoglu et al evaluated the antimicrobial effects of olive leaf extracts (aqueous, acetone, ethanol, and ethyl acetate) against some molds (having a key role in the spoilage of foods and beverages, primarily those with high acidity and reduced water activity) such as Saccharomyces cerevisiae ATCC 9763, Schizosaccharomyces pombe, Saccharomyces uvarum, Metschnikowia fructicola, Kloeckera apiculata, and Candida oleophila.
Acetone extract has been found most effective against Saccharomyces uvarum and Candida oleophila as acetone is the most effective solvent for dissolving dicarboxylic phenolic content of the olive leaf. None of the extracts exhibited antifungal activity against S. cerevisiae, except ethyl acetate. M. fructicola was found the most sensitive yeast to ethyl acetate extract followed by K. apiculata. S. cerevisiae demonstrated higher resistance than the other tested yeasts while S. pombe and C. oleophila were the most sensitive microorganisms [13].
Mycoplasma pneumonia, as well as at least 7 other mycoplasma species, have now been linked as a significant co-factor and as a direct cause of too many chronic diseases including, Alzheimer's, rheumatoid arthritis, multiple sclerosis, chronic fatigue, fibromyalgia, diabetes, Crohn's certain cancers, leukemia, etc. The activity of oleuropein was investigated in vitro against Mycoplasma fermentas, M. hominis, M. pneumoniae, and M. pirum. Oleuropein inhibited mycoplasmas at concentrations ranging from 20 to 320 mg·L−1. Despite the higher MIC values oleuropein has attracted considerable attention as a potential new natural drug for the treatment of infections due to mycoplasmas. Oleuropein has also shown activity against M. hominis and M. fermentans strains, which are naturally resistant to erythromycin and frequently to tetracyclines. The antimycoplasmal activity of oleuropein has been correlated with the o-diphenol system present on the oleuropein backbone structure [14].
Olive Leaf's Mechanism of Antifungal Action
The oleuropein and other polyphenolics mostly induce apoptosis in fungal cells as a primary mode of cell death as confirmed by the morphological changes in the nuclei after staining with fluorescent DNA-binding dyes. The antifungal agent targets virulence factors essential for the establishment of the fungal infection. Oleuropein modulates morphogenetic conversion and inhibits filamentation (as hyphae form stimulates virulence and plays an important role in tissue invasion and resistance to phagocytosis) of C. albicans.
The oleuropein has significantly decreased the cellular surface hydrophobicity (CSH) of C. albicans, a factor associated with adhesion to epithelial cells in both aerobic and anaerobic conditions. It also inhibits the activity of secreted aspartyl proteinases (SAP), cellular enzymes (related to the pathogenicity of the fungi) secreted by C. albicans. Furthermore, the oleuropein also causes a reduction in total sterol content in the membrane of C. albicans cells, which is involved in the mechanism of its antifungal activity [15].
Does Olive Leaf Have Antiviral Activity?
Olive leaf extract has been used in holistic medicine to treat definite viruses and exhibit antiviral properties. Oleuropein has shown potent antiviral activities against the hepatitis virus, herpes mononucleosis, rotavirus, canine parvovirus, bovine rhinovirus, and feline leukemia virus. Oleuropein exhibits considerable antiviral activity against parainfluenza type 3 virus, HIV, and respiratory syncytial virus.
This research has been limited only to cell cultures and has not been proven in human studies, but strong anecdotal evidence suggests it can effectively be used in the treatment of cold sores. The olive leaf extracts have also shown activity against viral hemorrhagic septicemia virus (VHSV), a salmonid rhabdovirus, and HIV-1 infection and replication [16, 17].
Olive Leaf's Other Health Benefits
Besides the antimicrobial activity of olive leaf extract, other several health benefits, have also been observed such as antioxidant, anticancer, anti-inflammatory, anti-hypertensive, and hypocholesterolemic properties. It was found that the ethanolic extract of olive leaves reduced blood glucose as a result of the increase in peripheral intake of glucose and insulin secretion induced by glucose in diabetic rabbits. The olive leaf extract increases glucagon-like peptide-1 secretion in vitro and in vivo environment and hence can be used for nutrition treatmentin Type-2 diabetes [18].
Olive leaf extract also exhibits hypolipidemic action via positively affecting atherosclerosis by decreasing low-density lipoprotein (LDL) and total cholesterol levels in rats alike atorvastatin [19].
Oleuropein enhances the nitric oxide (NO) production via induction of the inducible form of the enzyme nitric oxide synthase in macrophages challenged with lipopolysaccharide, thus increasing the functional activity of these immunocompetent cells.
Oleuropein elicits anti-inflammatory effects by the production of leukotriene B4 and inhibition of the lipoxygenase activity [20]. The high levels of oleuropein in olive leaf extract also act as a skin protectant, by acting as a free radical scavenger at the skin level, along with an anti-aging agent by decreasing intracellular levels of reactive oxygen species (ROS) and reduce the number of oxidized proteins through increased proteasome (a multicatalytic nonlysosomal protease and its increased expression delays senescence in human fibroblasts) mediated degradation rates and retain proteasome function during replicative senescence [21].
Suggested Olive Leaf Doses
Olive leave comes in soft gels, capsules, and tinctures. As such no recommended daily dose for olive leaf extract has been given. But the daily dose of 500 mg to 1,000 mg is considered to be safe. The splitting of the dose, from 250 mg to 500 mg two to four times a day with food has been recommended by most of the supplements.
Olive Leaf Side Effects
The Food and Drug Administration (FDA) considers olive leaf extract as a dietary supplement rather than a medicine. Hence they do not monitor the dose or quality of herbs and supplements. Most of the reported studies have shown that the olive leaf extracts are generally reliable and do not exhibit toxic effects even at high doses. The supplementation of olive leaf extract in female and male rats at a single dose of 2 gm/kg (acute toxicity) and 100, 200, and 400 mg/kg doses (subacute toxicity) given for 28 days did not result in any toxicity. Water-soluble extract of olive leaf was given at the doses of 360, 600, and 1,000 mg/kg/day for 90 days and did not cause any mortality and toxicity.
Also, there is no adequate evidence about its genotoxic effects in the literature. One study suggests that possible side effects may include stomach pain, muscle discomfort, and headache. Allergic reactions have also been developed due to the pollen from the olive leaf in some people. Thus, more studies are needed to fully understand the safety of olive leaf extracts for humans. The interactions of olive leaf extract with other medications have also not been fully explored. Hence persons who take prescription medications should speak to a doctor before taking the olive leaf extract [22,23].
To conclude:
Olive is a sacred and vital medicinal plant and the use of olive leaves can lower the risk of microbial infections, mainly in the respiratory, urinary, and intestinal tract primarily due to the antimicrobial action exhibited by the phenolic compounds present in it.
The use of olive leaf extracts is also suggested to achieve other health benefits like proven dermo-protective and therapeutic properties such as hypolipidemic anti-neoplastic, gastroprotective, anti-inflammatory, and anti-aging due to the additive and synergistic effects of phytochemicals present in the whole extract.
Additionally, it is non-toxic, non-hazardous, and completely safe. The outcome of already described phytochemical studies of olive leaves further expanded their therapeutic potential of olive leaves for more pharmacological studies, especially in vivo studies.
About the Author

Dr. Harmeet Kaur received her Bachelors in Pharmacy from Guru Nanak Dev University in Amritsar, India in 2000. Guru Nanak Dev University is a state owned university with an "A" grade nationally.
Dr. Kaur received her Masters in Medicinal Chemistry from the National Institute of Pharmaceutical Education and Research in 2002.
In 2015 Dr. Kaur was awarded her Ph.D in Pharmaceutical Sciences from Maharshi Dayanad University in Rohtak, India.
Dr. Kaur is presently a Senior Research Scientist at Maharshi Dayanand University in India.
Dr. Kaur has over 35 published Research papers concerning infectious diseases caused by yeasts, fungi, and bacteria using both prescription drugs and natural plant compounds. She has also performed many studies on cancer cells.
Of particular importance to us, is her multiple experiments performed on Candida albicans and pathogenic bacteria using natural compounds. Because of this experience, we are thrilled to have her on the YeastInfectionAdvisor team.
Back to Herbal Yeast Infection Remedies
Any questions about olive leaf or yeast infections in general, please feel free to contact us using the form on the contact page of this website or see your doctor.
Dr. Kaur's Medical References
[1]. Sun W, Frost B, Liu J. Oleuropein, unexpected benefits!. Oncotarget. 2017;8(11):17409.
[2]. Hassen I, Casabianca H, Hosni K. Biological activities of the natural antioxidant oleuropein: Exceeding the expectation–A mini-review. Journal of Functional Foods. 2015;18:926-940.
[3]. Hashmi MA, Khan A, Hanif M, Farooq U, Perveen S. Traditional uses, phytochemistry, and pharmacology of Olea europaea (olive). Evidence-Based Complementary and Alternative Medicine. 2015;2015.
[4]. Omar SH. Oleuropein in olive and its pharmacological effects. Scientia pharmaceutica. 2010;78(2):133-154.
[5]. Pereira AP, Ferreira IC, Marcelino F, Valentão P, Andrade PB, Seabra R, Estevinho L, Bento A, Pereira JA. Phenolic compounds and antimicrobial activity of olive (Olea europaea L. Cv. Cobrançosa) leaves. Molecules. 2007;12(5):1153-62.
[6]. Liu Y, McKeever LC, Malik NS. Assessment of the antimicrobial activity of olive leaf extract against foodborne bacterial pathogens. Frontiers in microbiology. 2017;8:113.
[7]. Sudjana AN, D’Orazio C, Ryan V, Rasool N, Ng J, Islam N, Riley TV, Hammer KA. Antimicrobial activity of commercial Olea europaea (olive) leaf extract. International journal of antimicrobial agents. 2009;33(5):461-463.
[8]. Soni MG, Burdock GA, Christian MS, Bitler CM, Crea R. Safety assessment of aqueous olive pulp extract as an antioxidant or antimicrobial agent in foods. Food and chemical toxicology. 2006;44(7):903-15.
[9]. Fleming HP, Walter WM, Etchells J. Antimicrobial properties of oleuropein and products of its hydrolysis from green olives. Applied microbiology. 1973;26(5):777-782.
[10]. Korukluoglu M, Sahan Y, Yigit A. Antifungal properties of olive leaf extracts and their phenolic compounds. Journal of food safety. 2008;28(1):76-87.
[11]. Zorić N, Kopjar N, Bobnjarić I, Horvat I, Tomić S, Kosalec I. Antifungal activity of oleuropein against Candida albicans-the in vitro study. Molecules. 2016 Dec;21(12):1631.
[12]. Nasrollahi Z, Abolhasannezhad M. Evaluation of the antifungal activity of olive leaf aqueous extracts against Candida albicans PTCC-5027. Current Medical Mycology. 2015;1(4):37.
[13]. Korukluoglu M, Sahan Y, Yigit A, Karakas R. Antifungal activity of olive leaf (Olea Europaea L.) extracts from the Trilye region of Turkey. Annals of microbiology. 2006;56(4):359.
[14]. Furneri PM, Marino A, Saija A, Uccella N, Bisignano G. In vitro antimycoplasmal activity of oleuropein. International journal of antimicrobial agents. 2002;20(4):293-296.
[15]. Zorić N, Kopjar N, Bobnjarić I, Horvat I, Tomić S, Kosalec I. Antifungal activity of oleuropein against Candida albicans-the in vitro study. Molecules. 2016;21(12):1631.
[16]. Fredrickson WR, inventor; F&S Group Inc, assignee. Method and composition for antiviral therapy. United States patent US 6,117,844. 2000 Sep 12.
[17]. Micol V, Caturla N, Pérez-Fons L, Más V, Pérez L, Estepa A. The olive leaf extract exhibits antiviral activity against viral haemorrhagic septicaemia rhabdovirus (VHSV). Antiviral research. 2005;66(2-3):129-136.
[18]. Acar-Tek N, Ağagündüz D. Olive Leaf (Olea europaea L. folium): Potential Effects on Glycemia and Lipidemia. Annals of Nutrition and Metabolism. 2020;76(1):63-68.
[19]. Olmez E, Vural K, Gok S, Ozturk Z, Kayalar H, Ayhan S, Var A. Olive leaf extract improves the atherogenic lipid profile in rats Fed a high cholesterol diet. Phytotherapy Research. 2015;29(10):1652-1657.
[20]. Haloui E, Marzouk B, Marzouk Z, Bouraoui A, Fenina N. Hydroxytyrosol and oleuropein from olive leaves: Potent anti-inflammatory and analgesic activities. Journal of Food, Agriculture & Environment. 2011;9(3-4):128-133.
[21]. Katsiki M, Chondrogianni N, Chinou I, Rivett AJ, Gonos ES. The olive constituent oleuropein exhibits proteasome stimulatory properties in vitro and confers life span extension of human embryonic fibroblasts. Rejuvenation Research. 2007;10(2):157-172.
[22]. https://www.medicalnewstoday.com/articles/324878
[23]. Omer SA, Elobeid MA, Elamin MH, Hassan ZK, Virk P, Daghestani MH, Al-Olayan EM, Al-Eisa NA, Almarhoon Z. Toxicity of olive leaves (Olea europaea L.) in Wistar albino rats. Asian Journal of Animal and Veterinary Advances. 2012;7(11):1175-1182.
Home Privacy Policy Copyright Policy Disclosure Policy Doctors Store
Copyright © 2003 - 2025. All Rights Reserved under USC Title 17. Do not copy
content from the pages of this website without our expressed written consent.
To do so is Plagiarism, Not Fair Use, is Illegal, and a violation of the
The Digital Millennium Copyright Act of 1998.