- Home
- Herbal Remedies
- Gentian Violet
Gentian Violet for Yeast & Bacterial Infections
Posted 6/15/2025
Written By Pharmaceutical Scientist Dr. Harmeet Kaur, PhD
This article talks about Gentian Violet effects on yeasts, bacteria and parasite infections. Possible safety issues and side effects.
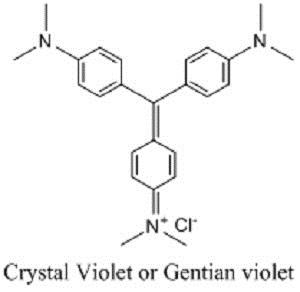
Gentian violet (GV; also known as crystal violet or methyl violet 10B) is a hexamethylpararosaniline chloride, a cationic triphenylmethane dye derived from aniline in which an amino group has been substituted by two methyl groups. It produces a deep violet-blue color, and the intensity of color depends on the number of methyl groups. The name gentian violet formerly used for the mixture of methyl pararosaniline dyes (methyl violet), but now it is denoted as crystal violet. The name signifies its color, which resembles the petals of some gentian flowers. It is used as a biological and histological dye, including as a critical stain in the Gram’s method for categorizing bacteria, as a nuclear stain, and as a commercial dye [1].
Gentian Violet History
In 1861, French chemist Charles Lauth synthesised GV under the name of ‘Violet de Paris’ and had it marketed by German pharmacist George Grubler [2,3].
In 1884 Hans Christian Gram observed the permanent fixation of GV by Gram-positive bacteria having the presence of peptidoglycan and lower lipid content, which became the basis of the Gram staining, one of the essential staining techniques for the categorization of the bacteria [4]. In 1891, Professor Stilling first introduced the antiseptic action of GV, which was marketed under the name of pyoctanin with various antimicrobial claims [5]. However, disputes arose about the curative allegations of pyoctanin resulted in the halt of further experimentation with GV in human subjects for the next two decades.
In 1912, Churchman reported the bacteriostatic potential of GV towards Gram-positive bacterial strains in vitro as well as in vivo in animals [6]. Up to the mid 20th century, GV has extensively been accepted for use in several diseases such as thrush, trench mouth, burns, impetigo, pinwormṣ, and systemic fungal infections. Due to varied composition of GV dyes, the claims of GVs efficacy were difficult to ascertain at that time and with the discovery and mass production of penicillin and sulfa drugs in the 1940s, physicians lost interest in the use of GV. The discovery of novel classes of antibiotics has become the central point of antimicrobial research [1].
Due to the crisis of antibiotic resistance nowadays, there has been a renewed interest in the GV therapy for anti-sepsis, along with some other uses particularly in developing countries due to its unique features like being easy to prepare, chemical stability, inexpensive, active at low concentration, broad-spectrum of antimicrobial activity with minimal resistance and less toxicity [1,7].
Gentian Violet as an Antimicrobial Agent
In 1992, Bakker et al. demonstrated GV as an anti-microbial for
dermatological disease. GV and brilliant green (a related
triphenylmethane dye), were applied to five bacterial species
(Staphylococcus aureus, Streptococcus A & B,
Pseudomonas aeruginosa, and Proteus) along with Candida
albicans in vitro. The study showed that in comparison to
brilliant green, GV was very active, exhibited low critical
concentration towards Streptococcus, Staphylococcus sp., and
Candida sp., and was moderately effective against the
Gram-negative bacteria (Pseudomonas sp. and Proteus)
[8].
GV has also shown its efficacy against methicillin-resistant
Staphylococcus aureus (MRSA). Saji et al. demonstrated the
complete eradication of MRSA from patients with MRSA infected
decubitus ulcers by using an ointment containing 0.1% GV
[9].
Further,
Bakker and Fung investigated the in vitro activity of GV
against Gram-negative bacteria particularly Pseudomonas, an
extremely virulent Gram-negative bacteria and found that GV
inhibited the growth of Pseudomonas, due to disruption
of Pseudomonas biofilms which are a vital source of
resistance to host defense and antibiotics [10].
Furthermore, medical equipment can also be protected from
microbial colonization by use of GV. Coating the equipment with
gendine, a mixture of gentian violet and chlorhexidine, of invasive
devices such as central venous catheters, urinary catheters and
endotracheal tubes have been shown to decrease the bacterial
adherence and prolong antimicrobial strength against the
Gram-negative organisms like E. Coli, Pseudomonas, and
Klebsiella both in vitro and in vivo
[11].
Gentian Violet for Oral & Breast Yeast Infections
Recently, GV has also shown its efficacy to treat oral candidiasis in human immunodeficiency virus (HIV) infected individuals particularly in developing countries where treatment with fluconazole is unfeasible due to its unavailability, treatment cost and the development of resistance. Typically, 0.5%-1% GV 1 – 2x/day is the dose used for mouth thrush in babies and adults having a weakened immune system. In breastfeeding women with thrush on the nipples, a 0.5% GV solution has been safely used for no longer than seven days [12,13].
Gentian Violet Activity Against Parasites
GV is also useful towards other infectious agents in addition to having antimicrobial properties. Since the 1950s, GV has been used safely in the prevention of transmission of Chagas’ disease (caused by protozoan parasite T. cruzi) as a blood additive in Latin America [14]. In a mouse model, GV has also shown its effectiveness both in vitro and in vivo towards the protozoa Leishmania mainly responsible for cutaneous leishmaniasis [15].
Gentian Violet Has Anti-tumor Activity
Recently, GV has also shown its efficacy as both anti-tumor and anti-angiogenic agent. In cell studies, GV has decreased the growth and killed the cancer cells of types such as colorectal, lung, breast, T-cell lymphoma, etc. However, no clinical trial has confirmed the use of GV for cancer [16].
Mechanism of Action and Interaction with Antimicrobial Agents
GV exert its effects via multiple mechanisms in bacteria, as being electrophilic during metabolic reactions it forms electrophilic metabolites like free carbon or nitrogen-centred radicals, formation of an un-ionized complex of bacteria with the dye, an alteration in redox potential by the dye, inhibition of reduced nicotinamide adenine dinucleotides phosphate (NADPH) oxidases, inhibition of essential amino acids and protein synthesis, uncoupling of oxidative phosphorylation, damage bacterial DNA, inhibition of formation of the bacterial cell wall, genotoxicity, and receptor-mediated effects.
Bacteria with powerful antioxidant mechanisms (anaerobic) or with a cell wall that does not allow the uptake of drugs easily (Gram-negative) can resist GV. In fungal strains such as Candida, GV inhibits spore maturation, and the synthesis of infection instigating harmful proteins. In the case of parasites causing Chagas’ disease, GV acts by increasing free radicals that damage it and inhibit protein production, energy, and calcium use in the parasite [17,18,19].
Gentian Violet Side Effects and Safety
GV is very chaotic; it stains the skin, teeth, and clothes, and almost anything else which comes into contact with it. Also, it can cause irritation or damage to the mouth and cheek lining and inflammation of the larynx when used for thrush. A high dose of GV (3%) has reportedly irritated the back of one person after 14 hours. Although rare, the repeated use of GV can cause allergic skin reactions. Workers exposed to high amounts (such as dye manufacturers, pulp workers, and fruit packers) have reported nose bleeding [20,21].
Gentian Violet Toxicity Issues
No severe toxicity has been reported when GV is used as a topical external treatment. Some controversy exists on its safety and cancer potential as GV can interact with the DNA of cells. However, GV is exceptionally safe and without significant contraindications for use. Despite over a century of use, GV has not directly linked to any cases of cancer. In humans, the toxicity of GV is limited only to case reports, and trials going on the usage of GV have demonstrated no or very mild adverse effects. The FDA also allows the sale of gentian violet over the counter [22].
To conclude, Gentian violet is an inexpensive drug having stability at room temperature for years. It is a drug that has a long history of medical use for various microbial infections. In many countries, it has become a staple of dermatologic treatment. When you consider the emergence of antibiotic resistance, the activity of GV at low concentration and minimal toxicity, the use of GV in the developed world would be helpful as well.
About the Author

Dr. Harmeet Kaur received her Bachelors in Pharmacy from Guru Nanak Dev University in Amritsar, India in 2000. Guru Nanak Dev University is a state owned university with an "A" grade nationally.
Dr. Kaur received her Masters in Medicinal Chemistry from the National Institute of Pharmaceutical Education and Research in 2002.
In 2015 Dr. Kaur was awarded her Ph.D in Pharmaceutical Sciences from Maharshi Dayanad University in Rohtak, India.
Dr. Kaur is presently a Senior Research Scientist at Maharshi Dayanand University in India.
Dr. Kaur has over 35 published Research papers concerning infectious diseases caused by yeasts, fungi, and bacteria using both prescription drugs and natural plant compounds. She has also performed many studies on cancer cells.
Of particular importance to us, is her multiple experiments performed on Candida albicans and pathogenic bacteria using natural compounds. Because of this experience, we are thrilled to have her on the Yeast Infection Advisor Team.
Back to Herbal Yeast Infection Remedies
If you have any questions about Gentian Violet, please feel free to contact us using the contact page of this website or talk to your doctor.
Dr. Kaur's References
1. Maley AM, Arbiser JL. Gentian Violet: a 19th century drug re‐emerges in the 21st century. Experimental dermatology. 2013; 22(12):775-80.
2. Lauth C. On the new aniline dye, Violet de paris. Laboratory. 1867; 1:138–139.
3. Titford M. George Grubler and Karl Hollborn: two founders of the biological stain industry. Journal of histotechnology. 1993; 16(2):155-8.
4. Gram C. Ueber die isolirte Farbung der Schizomyceten in Schnitt-und Trockenpraparaten. Fortschritte der Medicin. 1884; 2:185-9.
5. Stilling J. The anline dyes and antispetics. Lancet. 1891; 137:872–873.
6. Churchman JW. The selective bactericidal action of gentian violet. The Journal of experimental medicine. 1912; 16(2):221.
7. Fair RJ, Tor Y. Antibiotics and bacterial resistance in the 21st century. Perspectives in medicinal chemistry. 2014: PMC-S14459.
8.
Bakker P, Van Doorne H, Gooskens V, Wieringa NF. Activity of gentian
violet and brilliant green against some microorganisms associated with
skin infections. International journal of dermatology. 1992; 31(3):210-3.
9.
Saji M, Taguchi S, Hayama N, Ohzono E, Kobayashi Y, Uchiyama K,
Kanekawa T, Ohkuni H. Effect of gentian violet on the elimination of
methicillin-resistant Staphylococcus aureus (MRSA) existing in the
decubitus region. Nihon Ronen Igakkai Zasshi. 1993; 30:795–801. [PubMed: 8230793]
10. Fung DY, Miller RD. Effect of dyes on bacterial growth. Applied and environmental microbiology.1973 ;25(5):793-9.
11.
Chaiban G, Hanna H, Dvorak T, Raad I. A rapid method of impregnating
endotracheal tubes and urinary catheters with gendine: a novel
antiseptic agent. Journal of antimicrobial chemotherapy. 2005; 55(1):51-6.
12.
Nyst MJ, Perriens JH, Kimputu L, Lumbila M, Nelson AM, Piot P. Gentian
violet, ketoconazole and nystatin in oropharyngeal and esophageal
candidiasis in Zairian AIDS patients. Annales de la Societe Belge de
Medecine Tropicale.1992; 72:45-52. [PubMed: 1567268]
13.
Pienaar ED, Young T, Holmes H. Interventions for the prevention and
management of oropharyngeal candidiasis associated with HIV infection in
adults and children. Cochrane Database of Systematic Reviews. 2010(11).
14.
Nussenzweig V, Sonntag R, Biancalana A, et al. Effect of
triphenylmethane dyes on Trypanosoma cruzi in vitro; use of gentian
violet in prevention of transmission of Chagas disease by blood
transfusion. Hospital (Rio J). 1953; 44:731-744.
15.
de Souza Pietra RCC, Rodrigues LF, Teixeira E, et al. Triphenylmethane
derivatives have high in vitro and in vivo activity against the main
causative agents of cutaneous leishmaniasis. PloS one. 2013; 8:e51864.
16. Fried LE, Arbiser JL. Application of angiogenesis to clinical dermatology. Advances in dermatology. 2008; 24:89-103.
17.
Stearn EW, Stearn AE. The mechanical behavior of dyes, especially
gentian violet, in bacteriological media. Journal of bacteriology. 1923; 8(6):567.
18.
Ingraham MA. The bacteriostatic action of gentian violet and dependence
on the oxidation-reduction potential. Journal of bacteriology. 1933; 26(6):573.
19. Ying S, Qing S, Chunyang L. The effect of gentian violet on virulent properties of Candida albicans. Mycopathologia. 2010; 169(4):279-85.
20. John RW. Necrosis of oral mucosa after local application of crystal violet. British medical journal. 1968; 1(5585):157.
21.
Bielicky T, Novák M. Contact-group sensitization to triphenylmethane
dyes: gentian violet, brilliant green, and malachite green. Archives of
dermatology. 1969; 100(5):540-3.
22.
Littlefield NA, Gaylor DW, Blackwell BN, Allen RR. Chronic
toxicity/carcinogenicity studies of gentian violet in Fischer 344 rats:
two-generation exposure. Food and chemical toxicology. 1989; 27(4):239-47.
Home Privacy Policy Copyright Policy Disclosure Policy Doctors Store
Copyright © 2003 - 2025. All Rights Reserved under USC Title 17. Do not copy
content from the pages of this website without our expressed written consent.
To do so is Plagiarism, Not Fair Use, is Illegal, and a violation of the
The Digital Millennium Copyright Act of 1998.